A new way to treat disease
RNA editing has the potential to address the root causes of genetic diseases like alpha-1 antitrypsin deficiency (AATD).
Everyone is born with variations in genetic material — DNA and RNA — which serves as the blueprints for building the proteins that control cell function. Our company’s founders recognized that our bodies have a natural system for making changes to our RNA. By leveraging this system, we aim to reverse harmful genetic variations that cause missing or malfunctioning proteins, which can lead to serious diseases like alpha-1 antitrypsin deficiency (AATD).
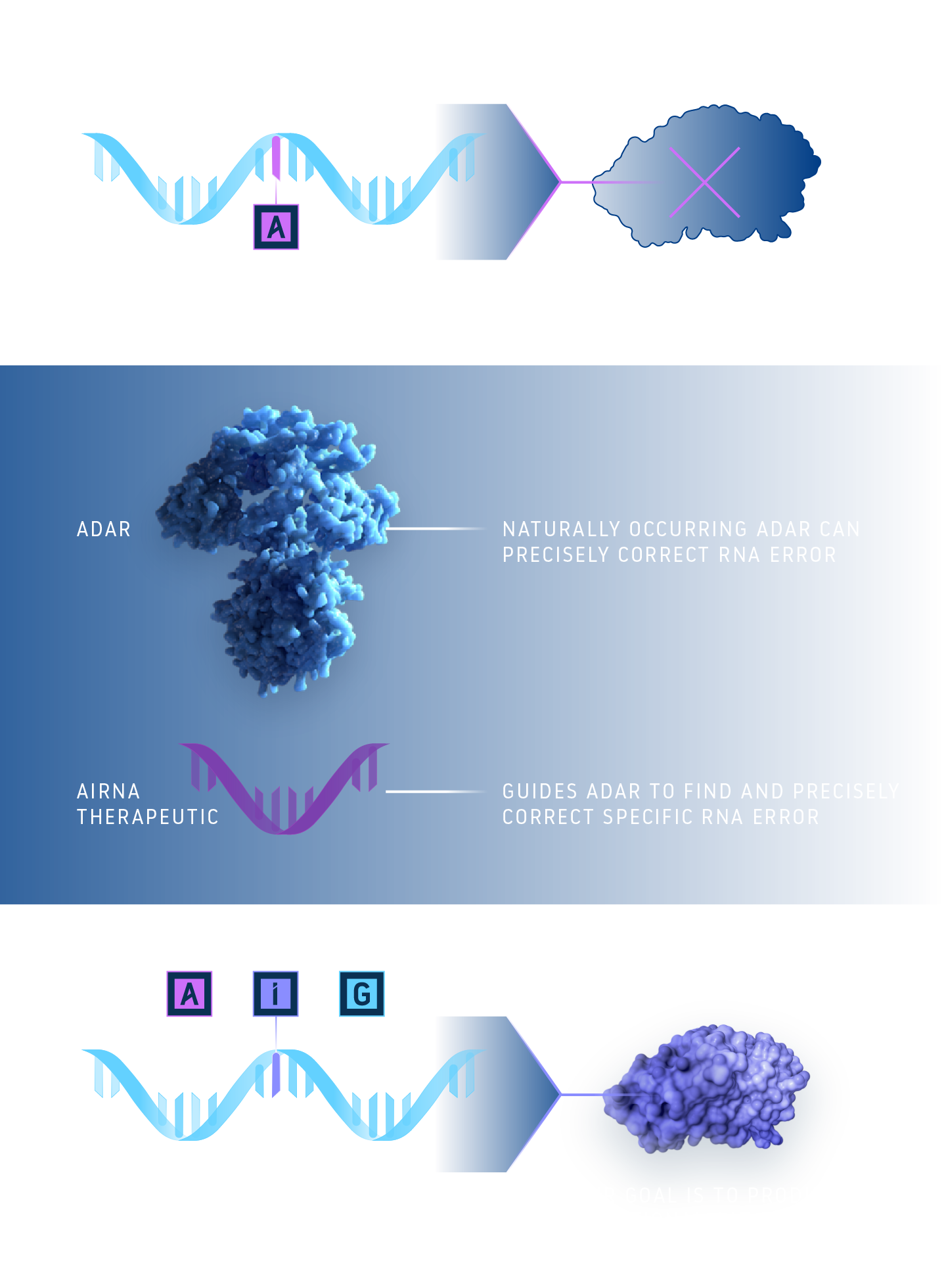
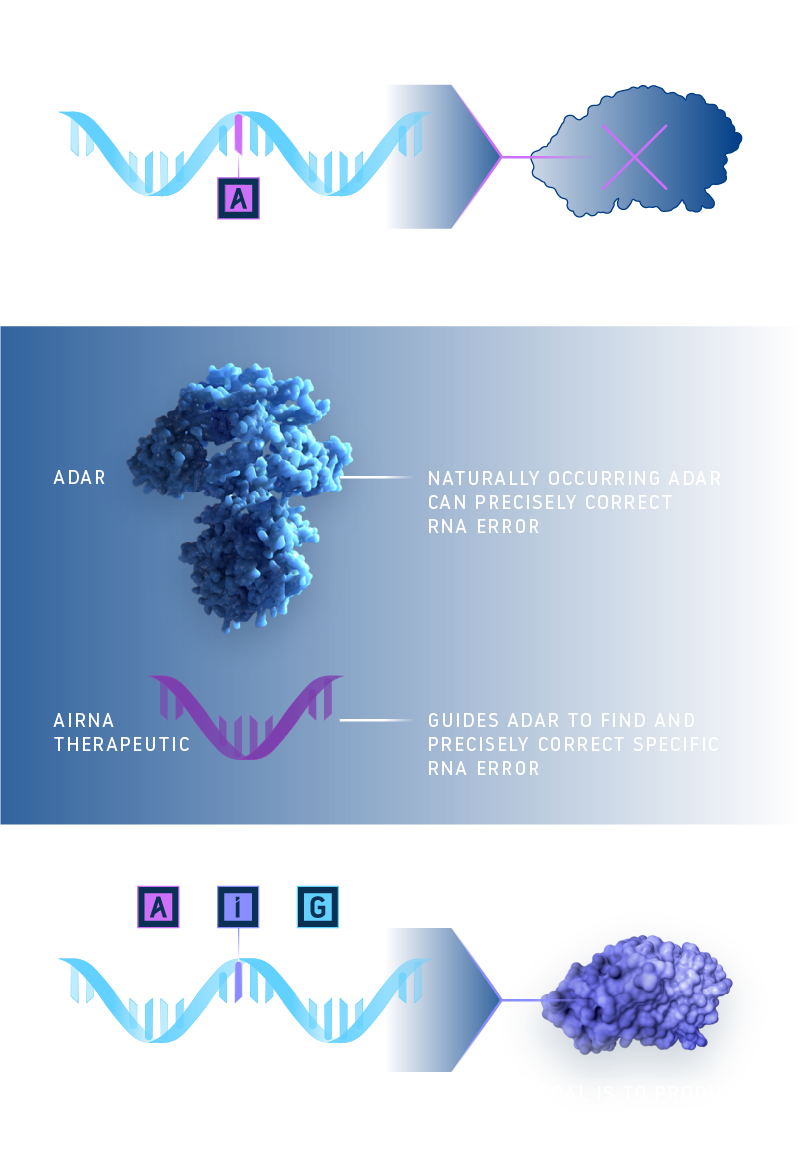
RNA is made up of a sequence of molecular subunits, often compared to letters in a code, that determine how proteins are built and function. The body’s natural RNA editing system uses a specialized protein called ADAR to make precise changes to the RNA by swapping one of the letters – switching an adenosine (A) to a new letter called an inosine (I), which is read as a guanosine (G) by the body’s genetic machinery. We are developing an investigational therapy that is designed to guide ADAR to make precise edits at the correct location. These small edits to RNA can lead to important improvements in how proteins function.
Harnessing a natural human process FOR AATD
AATD and precision RNA editing
Individuals with AATD have specific variations in their DNA and RNA that ultimately prevent production of functional alpha-1 antitrypsin (AAT) protein. AATD is inherited and can lead to severe lung and liver diseases, both of which currently lack sufficient treatment options.
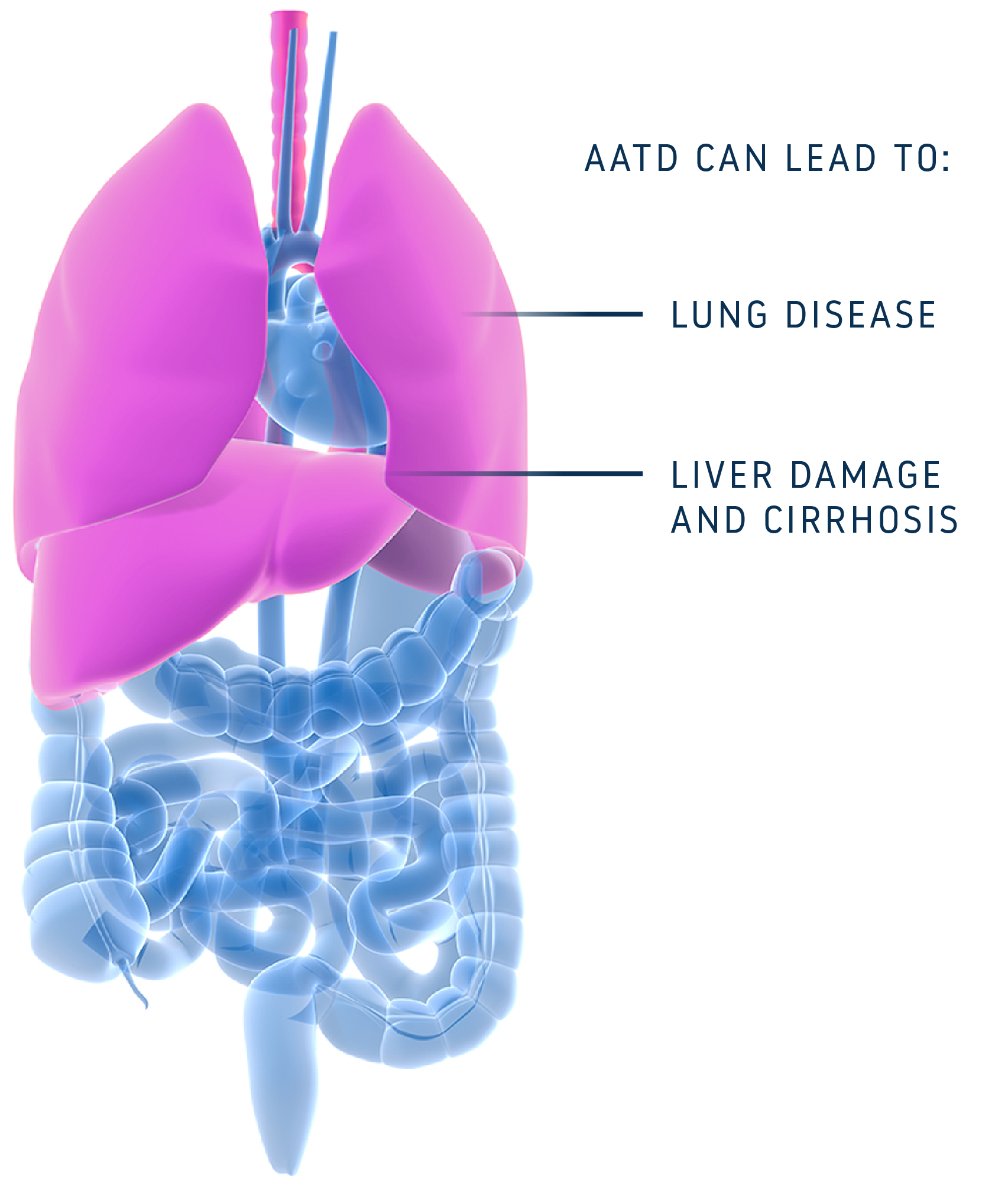
AIRNA’s precision RNA editing approach has the potential to naturally fix the most prevalent AATD-causing genetic variation (G-to-A) at the RNA level, leaving the DNA untouched but ensuring that the resulting protein will function properly. This approach avoids permanent risks.
AIR-001 was designed with the potential to provide a functional cure with a delivery profile preferred by patients and physicians. In preclinical research studies, AIR-001 potently repairs the RNA to produce high levels of AAT protein, with safe and convenient subcutaneous administration that is infrequently dosed.
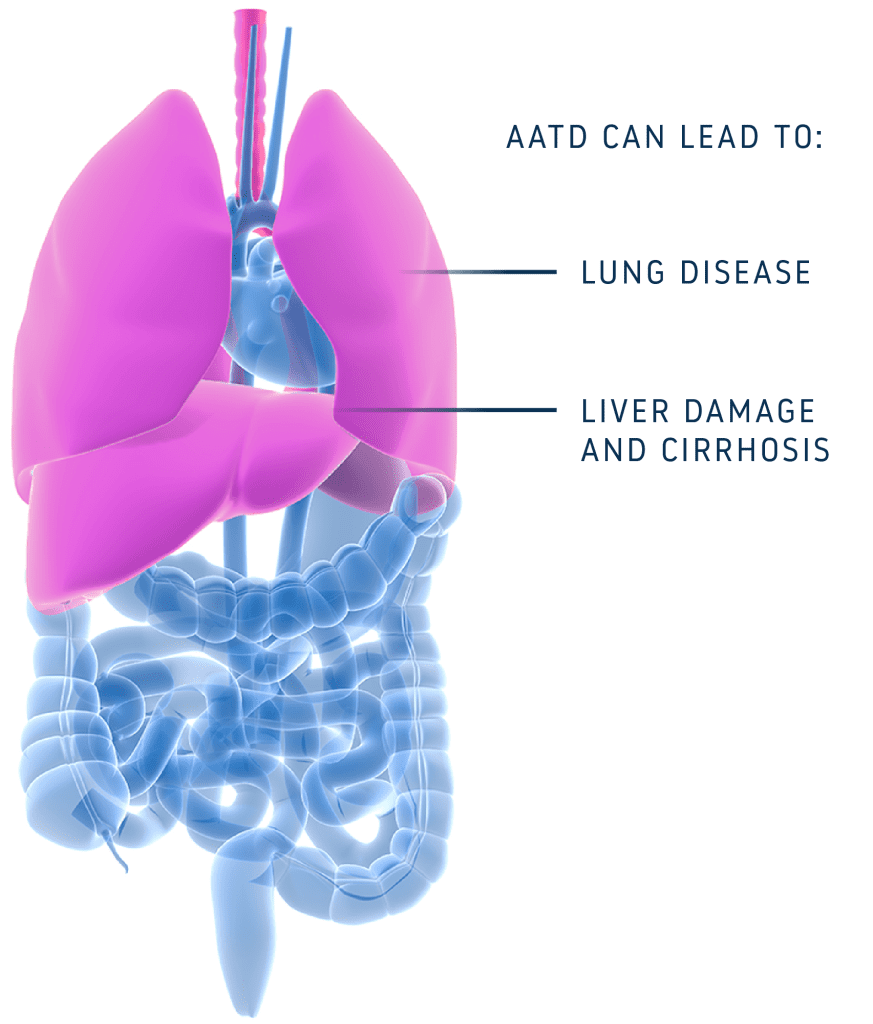
clinical trial updates
We are evaluating AIR-001 in the RepAIR1 Phase 1 global clinical study. RepAIR1 is an open-label, integrated single ascending and multiple dose study in adults with AATD who have the PiZZ genotype. It is designed to evaluate the safety, PK, and PD response to AIR-001, as assessed by changes in total AAT, functional AAT, Z-AAT and M-AAT.
Visit ClinicalTrials.gov to learn more about RepAIR1, or email patients@airna.com to sign up for clinical trial updates.

Learn more about AATD
For more information on living with AATD, explore the resources below:
Alpha-1 Europe Alliance
Alpha-1 Foundation
AlphaNet
American Lung Association
COPD Foundation
Uncover Alpha-1
